Chemically Critical
Glycol is a chemical commonly used in process cooling to reduce the temperature of materials for manufacturing. It’s used everywhere from lasers to dairy, and is often paired with water to hit freezing points well below zero. But don’t drink it!

Glycol: Keeping Things Cool
If you work with chillers in your industry, you likely have used glycol before. But what is it?
Chemical Uses
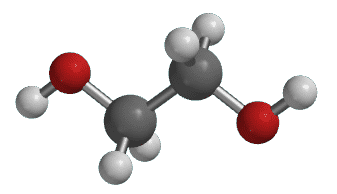
Glycol (HOCH2CH2OH) is a mildly toxic chemical that alters the freezing temperature of water. According to the American Chemistry Council, ethylene glycol (commonly referred to as glycol) is used for process cooling in many industries:
Ethylene glycol, a derivative of ethylene oxide, is used for the manufacture of polyester fiber for clothes, upholstery, carpet, and pillows and the blending of automotive engine antifreeze and coolant. Ethylene glycol is also used to manufacture fiberglass for products such as jet skis, bathtubs, and bowling balls. A major use is in the production of polyethylene terephthalate (PET) resin, a recyclable plastic, such as soda and water bottles.

Glycol and Water
When it comes to using glycol heaters or chillers, propylene glycol is the most important chemical when you have to balance cooling a product and preventing water from freezing. When mixed with glycol, water freezes at lower temperatures, allowing for cooler liquids to flow through a chiller system without the danger of the system freezing up.
The freezing levels of glycol by itself is 9°F. Mixing glycol with water lowers the new mixture’s freezing point, but that freezing point depends on the ratio of glycol to water. For example, an ambient temperature of -50°F can be reached with a 55% glycol, 45% water ratio.
Glycol in Chillers
Chillers use glycol-water mixtures to provide cooling to products and materials that need to be kept at a low, stable temperature. Glycol manufacturers such as Du Pont are required by law to provide a glycol level recommendation chart to consumers.
Learn More
Though not a glycol manufacturer, North Slope Chillers is an expert in the use of propylene glycol in chiller systems. For more information about glycol concentrations, click here.